

| Human iPS cells secrete SCGF (704, 710). Adenosine deaminase acting on RNA 1 (ADAR1) knockdown down-regulates several stem cell-related genes but up-regulates scgf gene in iPS cells derived from human foreskin fibroblasts (447). Multipotent adult progenitor cells (MAPCs) (30) MAPCs reside in the bone marrow, brain and muscle, and can differentiate into endo- (e.g. liver), meso- (e.g. endothelium) and ecto- (e.g. nerve) dermal lineage. Scgf gene is up-regulated 5-fold in the bone marrow and brain as compared with muscle (30). Mesenchymal stem cells (MSCs) (31, 32,244,252,265,387,512,638,Pat31) and fibroblasts Scgf gene is expressed in mouse BM stromal cells such as perivascular LepR+ MSCs and Col2.3+ osteoblasts (608, nichExplorer). Perivascular LepR+ MSCs include 4 clusters of Mgp+ P1, Lpl+ P2, Wif1+ P3 and Spp1+ P4, and Col2.3+ osteoblasts consist of 3 clusters of Col16a1+ O1, Fbn1+ O2 and Bglap+ O3. 5-FU-treated stress condition up-regulates scgf in Mgp+ P1, Wif1+ P3 and Spp1+ P4 clusters compared to steady state (608, nichExplorer). Scgf gene is highly expressed in an Alpl+Postn+ preosteoblast subset of mouse BM Cxcl12+LepR+ MSCs (614). Scgf-highly positive MSC subpopulation is predominantly present in human osteoblastic osteosarcoma tissues (638). 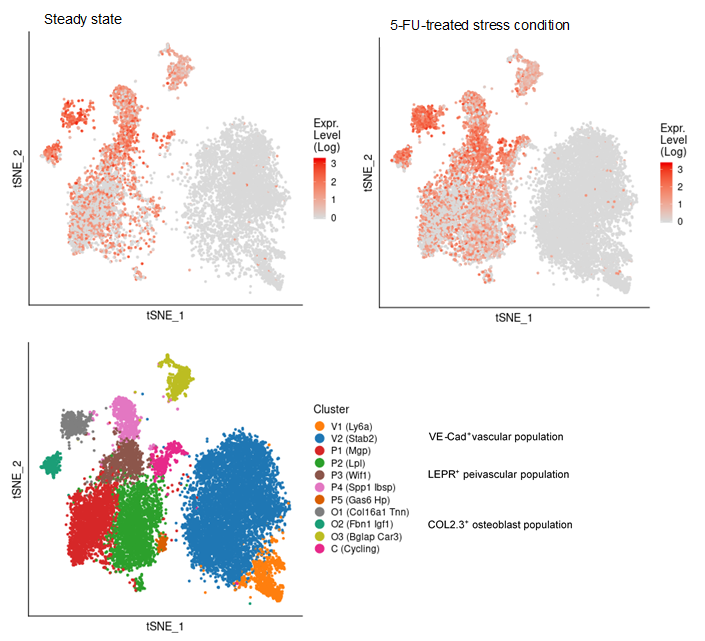 Human MSCs express scgf gene, but not G-, GM-, M-CSFs, ILs-6, 7, 8, 12, 14, HGF, LIF, flt3 L and c-kit L genes (31). It is controversial whether culture method of MSCs affects SCGF secretion. Human monolayer and spheroid MSCs secrete equal levels of SCGF (544), on the other hand, spheroid culture of human umbilical cord (UC)-derived MSCs considerably reduces their SCGF secretion relative to monolayer culture (575). Human monolayer UC-MSCs secrete higher level of SCGF in the vitreous fluid than spheroid UC-MSCs after transplanted into mouse vitreous (607). Human UC-MSCs are superior to BM-MSCs in secretory activity of SCGF (600). SCGF is highly enriched in human umbilical cord (UC)-MSCs-derived exosomes compared to parental MSCs. Human SCGF-rich UC-MSCs-derived exosomes increase trabecular bone mass, and decrease BM fat accumulation and osteoclasts when administered intravenously to mice with ovariectomy- or disuse (hindlimb-unloading by tail suspension)-induced osteoporosis (615). Human scgf+UC-MSCs-derived exosomes enhance and inhibit in vitro differentiation of mouse BM-MSCs into osteoblasts and adipocytes/osteoclasts, respectively, but human scgf- (silenced with scgfshRNA) UC-MSCs-derived exosomes exhibit no such bone effects. Differentiation of mouse Raw264.7 osteoclast progenitor cells into osteoclasts in the presence of RANKL is blocked by human scgf+but not scgf-UC-MSCs-derived exosomes. Mouse MSCs also express scgf gene in addition to BMP-4 and FGF-2 genes (32). FGF-2 is thought to be an elementary requisite for the growth of mouse MSCs, but not enough to maintain a long-term proliferation. Autocrine SCGF and BMP-4 may support MSC proliferation, while IGF, SCF and LIF are ineffective. However, SCGF is not the type of cytokine to stimulate MSCs to secrete IL-6 (33). Human adipose tissue-derived MSCs are successfully suspension cultured as self-assembling three dimensional spheroids in the defined medium containing SCGF, EGF, PDGF, TNF-α and IL-1β (369). MSCs from rat BM, adipose tissue and dental pulp commonly secrete SCGF, but BM and adipose tissue MSCs secrete much higher level of SCGF than dental pulp MSCs (512). Human microfragmented adipose tissues secrete higher levels of SCGF into the 14-28 day conditioned medium than lipoaspirate (595). Mouse but not human adipose tissue contains CD142+scgf+Fmo2+ preadipocytes that differentiate to Adipoq+Plin1+Car3+ adipocytes in response to insulin (602). An adipogenic differentiation of the CD142+scgf+Fmo2+ preadipocytes is moderately inhibited by TGF-β. Mouse Pdgfra+Bace2+Vipr2+scgf+ stromal cells reside close to adventitia of thoracic aorta in the perivascular adipose tissue (648). Human gingival papilla-derived MSCs also secrete SCGF (565). Scgf gene expression is highly down-regulated in the RelA-/- (a component of NF-κB) hES cell-derived MSCs, and they exhibit defective self-renewal ability (592). Knockdown of wild type hMSCs with scgf shRNA abrogates their proliferative potential, indicating that scgf is a mandatory driver for MSC proliferation. Scgf gene expression is up- and down-regulated in response to IL-2 in the self-renewal (0-40 day culture) and senescent (100-160 day culture) human adipose tissue-derived MSCs, respectively (507). SCGF protein resides in the exosomes from human (417, Exosome Database ExoCarta and Vesiclepedia), mouse (Exocarta) and rat (Exocarta) MSCs. Proteins involved in the exosome are listed at Exocarta, Vesiclepedia and Contents of the Exosome protein set, EMBL-EBI. Exogenous SCGF inhibits exosome production by human adipose tissue-derived MSCs (Pat35). Scgf gene expression is down-regulated along differentiation of adipose tissue MSCs to fat cells (277). Hypoxia-mimetic CoCl2 highly facilitates Simpson-Golabi-Behmel syndrome preadipocytes to secrete SCGF (430). Hypoxia also facilitates human MSCs to secrete SCGF during recovery after mechanical scratching stress (446). Wnt (wingless & int-related protein) gene has a commitment to the fate of stem cells in fetal tissues. Of wnt, wnt5a gene is specifically up-regulated in the osteoblasts differentiated from MSCs. Scgf gene is down-regulated in the calvaria of wnt5a-/- mice as compared with that of wnt5a+/+ mice (43). SCGF closely interacts with wnt5a in osseous differentiation. Transduction of mouse BM stromal OP9 cells with wnt3a alters their MSC characteristics into that of osteoblasts, and extremely up-regulates scgf gene expression in addition to decorin, fibromodulin and tenascin (361). When human adipose MSCs are cultured in the plate containing no osteogenesis-inducing factors, with or without microarc calcium phosphate (CaP)-coated titanium, they are differentiated to osteoblasts only in the plate with CaP-coated titanium (631). SCGF level is not different between both culture systems. SCGF has been thought to bind integrins (ITGs) through RGD motif sequence. Of human ITG α4β1, α9β1, α10β1, α11β1, αVβ1, αVβ3, αIIbβ3 and αMβ2 tested, human SCGF selectively binds ITGα10β1 and ITGα11β1 with similar high affinity as human pro-collagen 1α through RGD (594). Mouse SCGF acts on human and mouse MSCs to induce osteogenic differentiation despite lacking RGD through activated Wnt signal transduction, i.e. phosphorylation of GSK3, accumulation of nuclear β-catenin and up-regulation of Wnt target genes of Alp, Axin2, Lef1 and Runx2. ITGα10β1 and ITGα11β1 are not RGD-recognizing integrins and mouse SCGF lacks RGD, indicating that SCGF binds integrins through other mechanisms than RGD. Male mouse embryo fibroblasts (MEFs) express scgf gene 4 times higher than female MEFs in wild type mice, and the male-dependent scgf gene expression is down-regulated to the female level in KAI1/CD82-/- mice (465). Scgf gene expression is markedly up-regulated in the regenerating forelimb blastema of plethodontid salamander (Bolitoglossa ramosi) 40 days after amputation (590). Hematopoietic stem cells (HSCs) (34-39, 676) and osteoblasts (11, 40-44) or chondroblasts (11, 45, 46) Primitive hematopoietic stem/progenitor cells differentiated in vitro from H1 human ES cells express scgf gene (676). Interaction of HSCs with endosteal osteoblasts/chondroblasts is typical for that between stem cells and stem cell niche cells (47), the latter of which elaborate some mediators to support the former growth without any evidence for reverse signals from the former to the latter. Scgf gene expression is up-regulated in HOXB4-overexpressed c-kit+Lin-Sca-1+ subpopulation of murine EML primitive hematopoietic cells relative to GFP-expressing vector only EML cells, indicating that scgf is one of the "stemness"-specific target genes of HOXB4 (315). Scgf gene expression is rapidly and exceedingly up-regulated in the mouse CD45- stromal cells by 3 days after hematopoietic stem cell transplantation in sublethally irradiated host (396). Methylome analysis on hematopoietic progenitors indicates that CpG sites in the scgf promoter are differentially methylated between Lin-Il7ra-c-Kit+Sca-1+CD34+Flt3+ multipotent progenitors (MPP) and Lin-CD27+Flt3+Il7ra+Ly6D- common lymphoid progenitors (CLP) (MPP>CLP), between CLP and Lin-CD4-CD8-c-Kit+CD44+CD25-thymocyte progenitors (DN1) (DN1>CLP), and between DN1 and Lin-CD4-CD8-c-Kit+CD44+CD25+ thymocyte progenitors (DN2) (DN1>DN2) (284). Scgf gene expression is down-regulated in Dnmt3a-knockout (KO) mouse HSCs but up-regulated in Dnmt3a/Dnmt3b-double KO HSCs, while most genes show no dissociated regulation between Dnmt3a-KO and Dnmt3a/Dnmt3b-double KO HSCs (471). Dnmt3a/Dnmt3b-double KO HSCs further accelerate self-renewal with impaired differentiation seen in Dnmt3a-KO HSCs. Scgf gene expression is down-regulated along erythroid differentiation from CD45+GPA-IL-3R-CD34+CD36-CD71lo BFU-E to CD45+GPA-IL-3R-CD34-CD36+CD71hi CFU-E, and lost in the proerythroblasts (478). Mammary stem cells (48) and fat pad cells (49) Scgf gene is highly down- and up-regulated in CD29hiCD24+mammary stem cells from ovariectomized and pregnant mice, respectively (254). Oct4-transduced normal mammary epithelial cells up-regulate scgf gene expression and acquire cancer stem cell nature (345). Epidermal stem cells (352) Scgf is one of signature genes of dermal fibroblasts in the niche harboring embryonic hair follicle stem cells (Hair-GEL: A Transcriptome Atlas of Embryonic Hair Follicle Stem Cells and Their Niche, Icahn School of Medicine at Mount Sinai). When mouse live harvested dermal fibroblasts are cultured, scgf gene expression is significantly down-regulated compared to the level before culture (485). Circadian clock Per1 gene maintains murine CD34+Α6 integrinbrightPer1-venusbright hair follicle epidermal stem cells dormant, where scgf gene expression is up-regulated relative to the Per1-venusdim stem cells (352). Germline stem cells Testicular germ cells (50) and somatic cells of the testis (51). Female germline stem cells (FGSCs) Scgf gene is expressed in mouse ovarian female germline stem cells (FGSCs) (680). Daidzein up-regulates scgf to phosphorylate Akt, and promotes viability and proliferation of FGSCs. Fibroblasts Suppression of spindle assembly checkpoint S/T kinase BUB1 or mitotic cohesin SMC1A induces whole chromosome instability in human primary fibroblasts IMR-90 and PD25 to create senescent cell model. They significantly up-regulate scgf gene expression and SCGF secretion. Scgf is not up-regulated in the cells treated with paraquat or bleomycin, and SCGF secretion is proportional to the number of non-diploid cells, indicating that scgf is specifically up-regulated by whole chromosome instability (529). SCGF is one of senescence-associated phenotypes (SASP), and also associated with mortality and morbidity (637). Secretome analysis demonstrates SCGF in the conditioned media of human foreskin fibroblasts stimulated with FGF2 and TGF-β (578). Mouse fibroblast L cell exosomes contain SCGF (553). Scgf gene expression is down-regulated in the fluoride-resistant mouse L929 cells relative to wild type cells (561). The data described above are ascertained from the studies using relatively pure cell population. Major organs or tissues positive for expression of scgf mRNA include brain, eyes, bone marrow, blood cells, bone, cartilage, tooth, heart, breast, vascular endothelium, uterus, placenta, ovary, prostate, testis and skin, each of which is colored in red in the upper left panel. There have been many reports concerning molecular signature specific for organs or tissues by the use of microarray, SAGE or proteomics. Those data are derived from whole cells including stem/progenitor cells and other parenchymal cells, therefore it is uncertain which of the cells really express scgf mRNA. It would not be an exaggeration to say that bone and bone marrow are 2 major organs expressing scgf mRNA, that are most rich in stem cells at various levels adjacent to abundant stem cell niche cells.  In situhybridization of scgf mRNA SCGF is histochemically detected in the cytoplasm of human spleen cells and IFN-γ-activated acute monocytic leukemia cell line, THP-1 (R&D Systems). 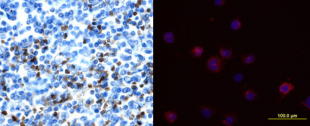 Cytoplasmic SCGF in human spleen cells (left) and IFN-γ-activated THP-1 cells (right) SCGF is histochemically detected in the cytoplasm of human hepatocellular carcinoma HepG2 cells, human kidney and brain tissue (Abcam). 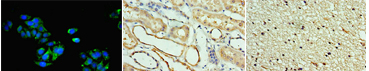 Cytoplasmic SCGF in human hepatocellular carcinoma HepG2 cells (left), human kidney (middle) and brain tissue (right) Various stem cells and cancer cells produce and secrete SCGF, and SCGF is usually detected in the cytoplasm and membranous region prior to release from those cells. Dynein transports SCGF along microtubules from cytoplasm into the pericentriolar material of centrosome. SCGF intriguingly plays a possible role in cell mitosis. 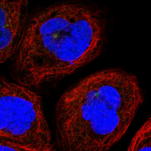 Centrosome is histochemically positive for SCGF in human A431 epidermoid carcinoma cells. (The Human Protein Atlas) Other cells, organs or tissues have been sporadically reported to express scgf mRNA, e.g. myoblast cell line, C2C12 (99, 100) [Scgf gene is down-regulated along with differentiation of mouse C2C12 myoblasts into myotubes (216).], pancreaticβ cells (101), colonic epithelial cell line, MCE301 (102), ileal enterochromaffin cells (103), cortical collecting duct of the kidney (104), fetal renal cells, HEK (105), fetal orofacial tissue (106), incisor enamel organ (107) and molar teeth (108). Scgf gene expression is 4.2-fold up-regulated along differentiation of chondroblasts and myofibroblasts in the culture of metanephroi with TGF-β2 (248). Scgf gene is significantly up-regulated in the tracheal explants 24hrs after exposure to cigarette smoke (251). LPS-stimulation of human adipocytes little affects their SCGF production (314). Scgf gene expression is highly up-regulated in the mouse peritoneal mesothelium but not in the pleural mesothelium relative to other tissue controls (342). Besides the expression data above based on the published papers, visit the following sites for more data from experiments and pathological specimen: GEO (Gene Expression Omnibus) at NCBI (National Center for Biotechnology Information): Over 2,000 results are presented. SymAtlas at Genomics Institute of the Novartis Research Foundation: CD34+ cells is prominent by far in expressing scgf gene. Expression Atlas at EMBL-EBI: Scgf gene is expressed predominantly in bone marrow. Genevisible at Genevestigator: BM cells, endothelial cells and neonatal fibroblasts are top-rank cells expressing scgf gene. GUDMAP (GenitoUrinary Development Molecular Anatomy Project): There are cumulative ISH and microarray data for scgf gene expression in developing and mature mouse gonads and urinary organ. Allen Institute for Brain Science: Accumulated are in situ hybridization images of scgf gene in mouse brain (coronal and sagittal) and spinal cord (juvenile and adult). Mouse Genome Informatics (MGI): Mouse scgf gene expression data are presented as an in situ hybridization image (62). Reference database for gene Expression Analysis (RefExA), System Biology and Medicine Database (SBM DB), Laboratory for System Biology and Medicien at RCAST, the Univeristy of Tokyo: Provides a heat map expression of scgf gene in a number of organs and cell lines. The Human Protein Atlas: Abundant histochemical staining data demonstrate SCGF in bone marrow subsets of hematopoietic cells, cerebellar Purkinje cells, hepatocytes, gall bladder glandular cells, pancreatic exocrineglandularcells, oral mucosal squamous epithelial cells, lung macrophages, post-menopausal uterine endometrial stromalcells, placental trophoblastic cells, epididymal glandular cells, kidney glomerular cells and urinary bladder urothelial cells. Subcellular localization database, COMPARTMENTS: SCGF protein is detected mainly in the extracellular region, but also subcellularly localized.
Cancer Cancer cells give rise to a totally different story about scgf mRNA expression from normal counterparts. Most fresh cancer cells or malignant cell lines established from cancer tissues uniformly up-regulate scgf gene expression. An expression of scgf gene might be attributable to an infinite self-renewal ability of cancer cells or cancer-initiating cells. Go see the Disease section for more information. Network between transcription factor, oncogene, histone modification and scgf Regulation of scgf gene expression by transcription factor, oncogene and histone modification is described in the Network section. |